Alzheimer’s disease (AD) is the most common neurodegenerative disease, clinically characterized by progressive memory loss. To date, an estimated 5.2 million people have the disease in the US, and the total number of people with AD-related dementia is projected to rise to 13.8 million by 2050.1,2 At present, there is no cure for the disease, and early clinical diagnosis is not yet available for the majority of patients.
The two main pathologic hallmarks of AD are senile plaques (amyloid plaques) and neurofibrillary tangles (NFTs), which develop in brain regions responsible for memory and cognitive functions (i.e. cerebral cortex and limbic system).3 Senile plaques are extracellular deposits of amyloid-β (Aβ) peptides, while NFTs are intracellular, filamentous aggregates of hyperphosphorylated tau protein.4
The identification of Aβ as the main component of senile plaques by Doctors Glenner and Wong in 19845 resulted in the original formation of the “amyloid hypothesis.” According to this hypothesis, which was later renamed the “amyloid-β cascade hypothesis” by Doctors Hardy and Higgins,6 the accumulation of Aβ is the initial pathologic trigger in the disease, subsequently leading to hyperphosphorylation of tau, causing NFTs and, ultimately, neuronal death and dementia.4,7–10 Although the details have been modified to reflect new findings, the core elements of this hypothesis remain unchanged: excess accumulation of the pathogenic forms of Aβ, by altered Aβ production and/or clearance, triggers the vicious pathogenic cascades that eventually lead to NFTs and neuronal death.
The Amyloid β Hypothesis—A Causal Link Between Amyloid β and Neurofibrillary Tangles?
Over the last two decades, the Aβ hypothesis of AD has reigned, providing the foundation for numerous basic studies and clinical trials.4,7,10,11 According to this hypothesis, the accumulation of Aβ, either by altered Aβ production and/or clearance, is the initial pathologic trigger in the disease. The excess accumulation of Aβ then elicits a pathogenic cascade including synaptic deficits, altered neuronal activity, inflammation, oxidative stress, neuronal injury, hyperphosphorylation of tau causing NFTs and, ultimately, neuronal death and dementia.4,7–10 However, the Aβ hypothesis has not been fully validated and several major unresolved matters remain.12–14 Recent failures of human clinical trials have raised concerns about whether blocking toxic Aβ accumulation is sufficient to stop and even reverse the progression of downstream AD pathologies.15–17
One of the major unresolved factors of the Aβ hypothesis is to show a direct causal link between Aβ and NFTs.12–14 Studies have demonstrated that treatments with various forms of soluble Aβ oligomers induced synaptic deficits and neuronal injury, as well as hyperphosphorylation of tau proteins, in mouse and rat neurons, which could lead to NFTs and neurodegeneration in vivo.18–21 However, transgenic AD mouse models carrying single or multiple human familial AD (FAD) mutations in amyloid precursor protein (APP) and/or presenilin 1 (PS1) do not develop NFTs or robust neurodegeneration as observed in human patients, despite robust Aβ deposition.13,22,23 Double and triple transgenic mouse models, harboring both FAD and tau mutations linked with frontotemporal dementia (FTD), are the only rodent models to date displaying both amyloid plaques and NFTs. However, the NFT pathology in these models stems mainly from the overexpression of human tau as a result of the FTD, rather than the FAD mutations.24,25
Failure of attempts at full recapitulation of AD pathologies in mice might be also due to fundamental species-specific differences between mice and humans. Indeed, adult mice do not express the six human isoforms of tau proteins and endogenous mouse tau seems to interfere with aggregation of human tau proteins.26
Testing Amyloid β Cascade Hypothesis in Human Neuronal Models
Recent reprogramming technology has provided a new model to test the amyloid hypothesis. Specifically, induced pluripotent stem cells (iPSCs) can be generated from the fibroblasts of AD patients harboring single FAD mutation either in APP or PS1.27–34 These neurons showed significant increases in the ratio of pathogenic Aβ42 to Aβ40 compared with non-AD control neurons.4,27,28,31,35–37 Human neurons with the APP duplication FAD mutation also displayed robust increases in total Aβ levels due to heightened levels of APP, the precursor protein for Aβ generation.27,33 Similarly, trisomy 21 (Down syndrome) neurons also showed robust increases in total Aβ levels due to the APP gene duplication, which is located on chromosome 21.38
Human neurons carrying FAD mutations are an optimal model to test whether elevated levels of pathogenic Aβ trigger pathogenic cascades including NFTs, since those cells truly share the same genetic background that induces FAD in humans. Indeed, Israel et al., observed elevated tau phosphorylation in neurons with an APP duplication FAD mutation.33 Blocking Aβ generation by β-secretase inhibitors significantly decreased tau phosphorylation in the same model, but γ-secretase inhibitor, another Aβ blocker, did not affect tau phosphorylation.33 Neurons with the APP V717I FAD mutation also showed an increase in levels of phospho tau and total tau levels.28 More importantly, Muratore and colleagues showed that
treatments with Aβ-neutralizing antibodies in those cells significantly reduced the elevated total and phospho tau levels at the early stages of differentiation, suggesting that blocking pathogenic Aβ can reverse the abnormal tau accumulation in APP V717I neurons.28
Recently, Moore et al. also reported that neurons harboring the APP V717I or the APP duplication FAD mutation showed increases in both total and phospho tau levels.27 Interestingly, altered tau levels were not detected in human neurons carrying PS1 FAD mutations, which significantly increased pathogenic Aβ42 species in the same cells.27 Treatments with β-secretase inhibitor significantly decreased phospho and total tau levels in the APP V717I and the APP duplication models, but γ-secretase inhibitor could not reduce abnormal tau accumulation in the same cells.27 These data suggest that elevated tau levels in these models were not due to extracellular Aβ accumulation but may possibly represent an early stage of tauopathy. Alternatively, it may result from developmental alterations induced by the APP FAD mutations. Further studies will be needed to clarify the pathogenic importance of tau changes in human iPSC-derived AD neurons.
One of the challenges of replicating tauopathy in human iPSC-derived neurons is that wild-type human iPSC-derived neurons, despite longer differentiation (>100 days), do not fully express adult tau splicing isoforms.39–41 The presence of select FTD tau mutations enhances the expression of adult 4-repeat tau splicing isoforms.39–41 While control wild-type neurons do not express adult tau isoforms in the same conditions.39–41 This clearly limits the recapitulation of human tauopathy, in which 4-repeat tau plays an important role, in human iPSC-derived neurons without FTD tau mutations.
As summarized, most human FAD neurons showed significant increases in pathogenic Aβ species, while only APP FAD neurons showed altered tau metabolism possibly representing early stages of tauopathy. However, all of these human FAD neurons failed to recapitulate robust extracellular amyloid plaques, NFTs, or any signs of neuronal death, as predicted in the amyloid hypothesis. Difficulty proving the amyloid hypothesis thus far in FAD iPSC neurons might be a consequence of the low levels of pathogenic Aβ in these cultures. Average Aβ levels in brains of AD patients are much higher than those achieved in FAD iPSC-derived neuronal cells.27–34,42 It is possible that human iPSC-derived FAD neurons may not be suitable for generation of elevated Aβ levels on par with levels found in the brains of AD patients.43
Modeling Amyloid Plaques and Neurofibrillary Tangles in a Human Neural 3D Culture System
In our recent study, we moved one step closer to proving the amyloid hypothesis. By generating human neural stem cell lines carrying multiple mutations in APP as well as PS1, we achieved high levels of pathogenic Aβ42 comparable to those in brains of AD patients.44–46 Co-expression of multiple FAD mutations in APP and PS1 has been previously employed for generations of AD transgenic mouse models. This strategy has been shown to increase aggregation-prone Aβ42 levels through both dramatic acceleration of onset and increased total levels of Aβ deposition.22,23,47
Secreted Aβ in a conventional 2D cell culture system was observed to diffuse into the cell culture media, and be removed during media changes, precluding any possibility of aggregation. This finding led us to adopt a novel 3D Matrigel® culture system to create an environment in which secreted Aβ accumulates, accelerating Aβ aggregation.44,45 After 6 weeks of differentiation
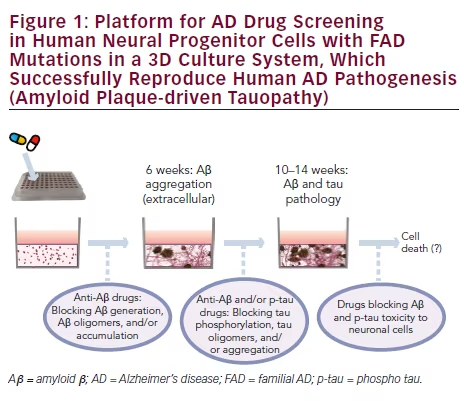
in our 3D Matrigel system, our FAD ReNcell VM human neural precursor cells showed robust extracellular Aβ deposits and detergent sodium dodecyl sulfate (SDS)-resistant Aβ aggregates (Aβ dimers, trimers, and tetramers).44,45 Importantly, we observed accumulations of hyperphosphorylated tau proteins in somatodendritic compartments, which were also present in detergent-insoluble fractions.44,45 Immunoelectron microscopy confirmed the presence of detergent-insoluble filamentous structures labeled by tau antibodies.44 Taken together, these observations clearly demonstrated the presence of Aβ plaques and NFT-like pathologies in our 3D human AD culture model. Notably, these AD pathologies were induced solely by FAD mutations without co-expressing human tau FTD mutations.
Although our 3D human cellular AD model was able to simulate major pathogenic events of AD, many challenges remain in comprehensive recapitulation. One major disadvantage of our model is manifested in the use of transgenic overexpression of APP and PS1 to generate elevated Aβ levels. This nonphysiologic overexpression of APP may lead to supplementary pathogenic effects in addition to the accumulation of Aβ, as observed in transgenic AD mouse models.48 Furthermore, the limitations in differentiating ReN cells into mature forebrain neurons, which are most affected in AD, may also pose a formidable challenge in fully reconstituting pathogenic cascades of AD.
Clinical Implication of Human Cellular Models of Alzheimer’s Disease
One of the major advantages of human cellular models of AD is that they can provide platforms for high-throughput screening (HTS) of new AD drugs in a human brain-like environment. Our studies clearly demonstrated the advantage of using human neural cell culture systems in recapitulating Aβ-induced tauopathy, which was not feasible in mouse models.44–46 Our 3D human cellular AD model can also provide a powerful platform for HTS of candidate AD drugs that can reduce Aβ and/or tau pathology in a single system, which is not possible in current AD models (see Figure 1). Also, our 3D culture systems allow scalable plating techniques, from thick-layer 6- and 24-well formats to a 96-well thin-layer design, which can fit largescale HTS or high-content screening (HCS).44–46 An additional strength of our immortalized and single-clonal human neural progenitor cells is their rapid proliferation and stability throughout repeated passages.
Another potential application of human cellular models of AD is to validate/ optimize current therapeutic approaches for AD in a human brain-like environment. For example, various antibodies against monomeric and/or oligomeric Aβ species have been under human clinical trials, mostly based on success in AD mouse models.49 As shown by Muratore et al., the efficacy of Αβ neutralizing antibodies can also be tested in a human neural cell culture model of AD.28 In addition to the impact on toxic Aβ species, our 3D culture model can test if these antibodies can block tau pathologies in 3D human neural cell culture systems.44–46 Human cellular AD models can also be used to determine optimal doses of candidate AD drugs to block Aβ and/or tau pathology without affecting neuronal survival (see Figure 1). Indeed, a recent study has shown that human iPSC-derived neurons are more resistant to nonsteroidal anti-inflammatory drug (NSAID)- based γ-secretase modulation compared with rodent neurons, which may explain the failure of human clinical trials of select NSAIDs.50
In addition to drug screening, human cellular AD models can be used to explore molecular mechanisms underlying AD pathogenesis, which could provide novel druggable targets to reduce AD pathology.51 While human iPSC-derived AD models allow study of pathogenic mechanisms under physiologic conditions, our 3D model provides insight after pathologic accumulation of β-amyloid and NFT formation, which are present in moderate to advanced AD.
While much progress has been made, many challenges still lie on the path to creating human neural cell culture models that comprehensively recapitulate pathogenic cascades of AD. A major difficulty lies in reconstituting the brain regions most affected in AD: the hippocampus and specific cortical layers. Recent progress in 3D culture technology, such as “cerebral organoids” may also be helpful in rebuilding the brain structures that are affected by AD in a dish.52,53 These “cerebral organoids” were able to model various discrete brain regions including human cortical areas,52 allowing them to reproduce microcephaly, a brain developmental disorder. Similarly, pathogenic cascades of AD may be recapitulated in cortex-like structures using this model. Adding neuroinflammatory components, such as microglial cells, which are critical in AD pathogenesis, will illuminate the validity of the amyloid β hypothesis. Reconstitution of robust neuronal death stemming from Aβ and tau pathologies will be the next major step in comprehensively recapitulating AD in a cellular model.
Another major challenge lies in developing a precise model for sporadic AD. Currently all of the AD mouse models harbor FAD mutations, which are able to reconstitute pathogenic mechanisms of FAD. Conversely, sporadic AD is caused by both genetic and environmental factors that have not been fully characterized yet. Human iPSC-derived neurons from sporadic AD patients
can provide a model for sporadic AD.32,33,54,55 Indeed, some human iPSC cell lines derived from sporadic AD patients showed elevated levels of Aβ.33,54 However, these Aβ increases have not been consistently observed in other sporadic iPSC-derived neurons.32,33 Also, no severe AD pathologies, including extracellular aggregation of β-amyloid or robust tauopathy, were observed in these cells.32,33,54,55 These inconsistencies clearly demonstrate the technical difficulties of modeling sporadic AD even with human iPSCs derived from patients. However, according to the Aβ hypothesis, sporadic AD and FAD share common pathogenic pathways. Thus, drugs that can block Aβ toxicity may work for both FAD and sporadic AD patients, which justifies the use of FAD models to develop drugs for sporadic AD patients.
We predict that further advances in human stem cell technology, as well as recent progress in 3D culture technology, will generate cellular AD models that can more precisely mimic AD pathogenesis in a dish. These AD models will accelerate discovery of new AD drugs and also enable dissection of molecular mechanisms underlying the pathogenic cascades of AD.







